- 3999
- 405
- 10
- 16
- 0
- Help Ukraine
About the solution
Brett Tripp, the owner of Tripp Design in Ann Arbor, Michigan, has pioneered an innovative project aimed at revolutionizing wearable medical devices for individuals managing Inflammatory Bowel Disease (IBD). Diagnosed with Crohn's disease at a young age, Brett's firsthand experience as a patient advocate inspired him to address the lack of customizable options in the market.
Utilizing his design expertise, Brett began by 3D printing custom ostomy wafers in his garage, a project that has since gained momentum. Collaborating with Dr. Higgins of the University of Michigan IBD Program, they have created six prototypes, each building upon the previous iteration. These prototypes are developed based on 3-D models generated from digital scans of ostomy sites and stomas of six patients, ensuring a precise fit and comfort.
One significant advancement in the project is the transition from conventional pectin-based wafers to a more durable medical-grade silicone material. This material not only offers flexibility, allowing patients to maintain a secure seal even during movements but also represents a significant improvement in durability.
Currently, Brett and Dr. Higgins are actively seeking sponsorship to further develop and enhance their project. Their goal is to make their custom ostomy wafers even better, offering individuals with IBD an improved quality of life. The impact of this project extends beyond innovation; it reaches into the daily lives of patients, highlighting the motivation behind their work. They are driven by the belief that they are "on to something" that can significantly improve the lives of individuals managing IBD, ultimately reinforcing the importance of innovation and empathy in healthcare.
Source of information: https://lyfebulb.com/people/brett-tripp/
For more information: https://twitter.com/Trippdesign | https://www.michiganmedicine.org/health-lab/how-3-d-printing-makes-ostom...
This solution shall not include mention to the use of drugs, chemicals or biologicals (including food); invasive devices; offensive, commercial or inherently dangerous content. This solution was not medically validated. Proceed with caution! If you have any doubts, please consult with a health professional.
DISCLAIMER: This story was written by someone who is not the author of the solution, therefore please be advised that, although it was written with the utmost respect for the innovation and the innovator, there can be some incorrect statements. If you find any errors please contact the patient Innovation team via info@patient-innovation.com
-
-
425
-
0
-
4323
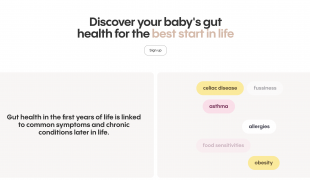
Nora Cavani and Willem de Vos founded Alba Health- a kit test- to help understand microbiome science and improve children's health.
(SELF)-CARE: DRINKING: Drinking independently.
(SELF)-CARE: EATING: Eating independently.
(SELF)-CONTROL: BOWEL CONTINENCE: Maintaining bowel continence
(SELF)-CARE: USING THE TOILET: Using the toilet independently
Allergic reaction (food, drugs, material/chemicals)
Online service
Skin redness (rash)
Itching (pruritus)
Urticaria
Skin blisters
Dry skin
Skin vesicles
Skin lumps or growths
Skin pustules
Hair loss (alopecia)
Skin ulcers or sores
Skin discoloration
Hair thinning or brittleness
Nail abnormalities
Nail brit (weak nail)
Photosensitivity
Acne
Skin thickening (lichenification)
Transpiration (Sweating)
Managing pain
Enhancing health literacy
Promoting self-management
Enhancing digestive function
Preserving Organ Function
Building Supportive Community Relationships
Promoting inclusivity and social integration
To implement a diagnostic tool
Alleviating Allergies
To improve Treatment/Therapy
Endocrinology
General and Family Medicine
Infectious Diseases
Immuno-allergology
Internal Medicine
Neurology
Otorhinolaryngology
Pediatrics
Plastic and Reconstructive Surgery
Sweden
-
-
-
426
-
0
-
3981
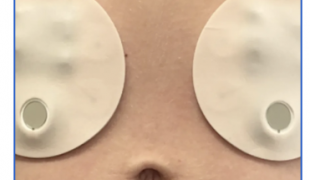
Caregiver Steve Axelrod creates EKG for the gut - a patch-based system is designed to read signals from the digestive tract.
(SELF)-CARE: HYGIENE: Maintaining personal hygiene
(SELF)-CARE: USING THE TOILET: Using the toilet independently
(SELF)-CONTROL: BOWEL CONTINENCE: Maintaining bowel continence
CAREGIVING
Chron's Disease
Inflammatory Bowel Disease
Assistive Daily Life Device (to help ADL)
Change in bowel habits
Bloating
Constipation or diarrhea
Promoting self-management
Enhancing digestive function
Preserving Organ Function
Restoring Skin Health
Alleviating Allergies
To improve Treatment/Therapy
Preventing (Vaccination, Protection, Falls, Research/Mapping)
Raise awareness
Caregiving Support
Gastroenterology
General and Family Medicine
General Surgery
Pediatrics
United States
-
-
-
419
-
0
-
4398

Patient Sam Jactel designed an app - Ayble health - that allows patients to create a personalised plan for a healthier gut.
(SELF)-CARE: EATING: Eating independently.
(SELF)-CARE: DRINKING: Drinking independently.
(SELF)-CONTROL: BOWEL CONTINENCE: Maintaining bowel continence
CAREGIVING
Cooking
Social interaction
Chron's Disease
oesophageal disorders (oesophagitis, gastroesophageal reflux disease, gastroesophageal sphincter insufficiency)
Inflammatory Bowel Disease
App (Including when connected with wearable)
Strategy/Tip
Abdominal pain
Weight loss
Indigestion (dyspepsia)
Vomiting (Regurgitation)
Abdominal swelling or mass
Weight gain
Enhancing health literacy
Promoting self-management
Enhancing digestive function
Manage Medication
Preserving Organ Function
Alleviating Allergies
Preventing (Vaccination, Protection, Falls, Research/Mapping)
Raise awareness
Caregiving Support
Gastroenterology
General and Family Medicine
General Surgery
Internal Medicine
Pediatrics
United States
-
 en
en